About RepliCel
Overview
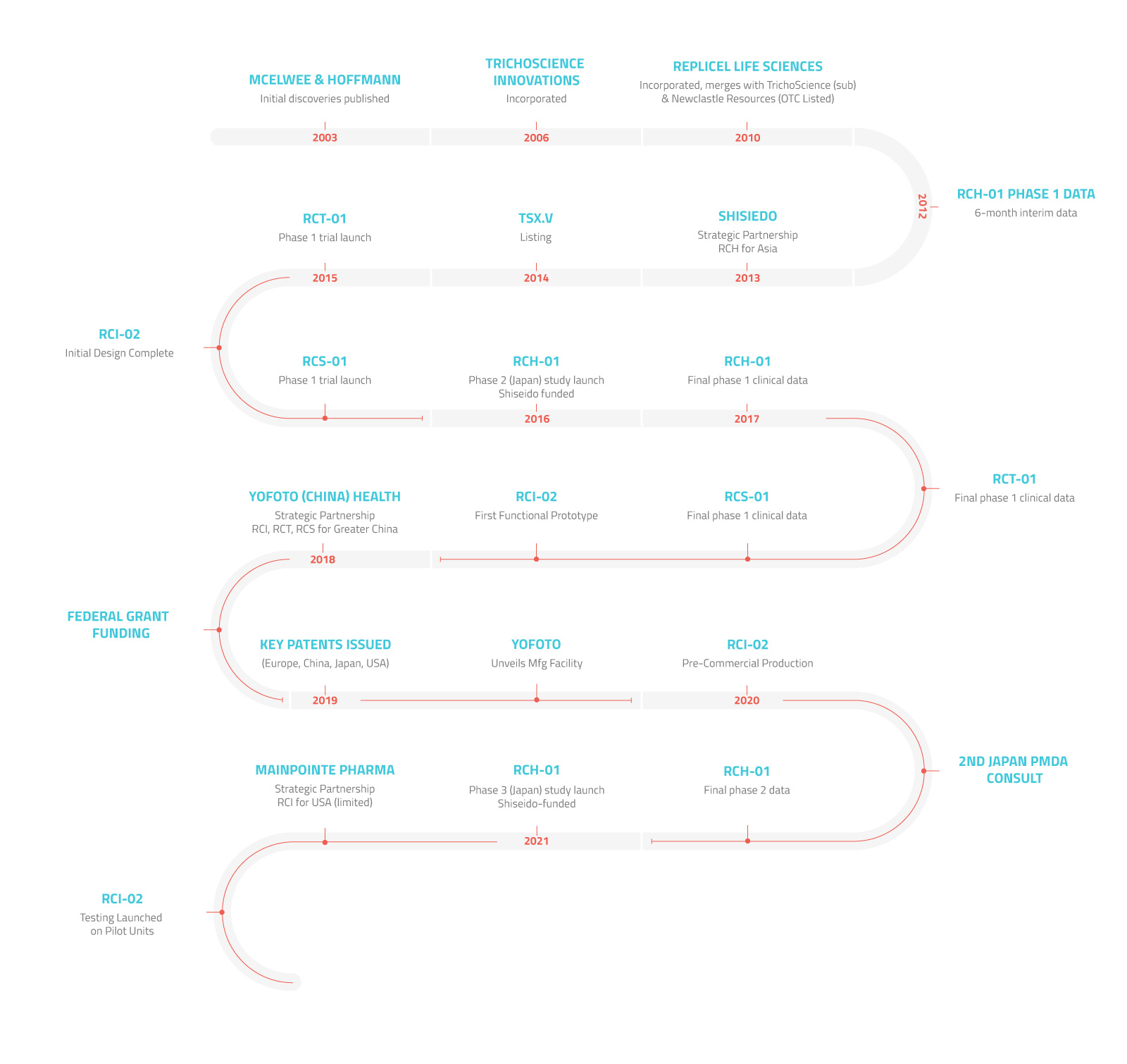
RepliCel is a regenerative medicine company focused on aesthetics and orthopedics. Our cell therapy products are tailor-made to restore cellular function to rejuvenate aging or sun damaged skin, regrow hair, and repair tendons. Our next-generation dermal injection technology is designed to revolutionize aesthetic treatment outcomes. RepliCel is dedicated to improving everyday lives.
Technology
RepliCel is developing several products from two unique cell therapy populations both taken from the hair follicles embedded in a small skin biopsy taken from the back of a patient's head.
RepliCel is also developing a medical device based on several patent-protected technologies related to improving patient injections.
Products
The products currently RepliCel is currently developing are:
- RCH-01 – Cell therapy for pattern baldness
- RCS-01 – Cell therapy for aging or sun damaged skin
- RCT-01 – Cell therapy for tendon degeneration
- DermaPrecise™ – Next-generation dermal injection device